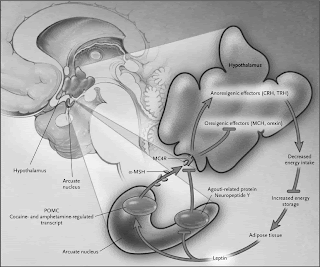
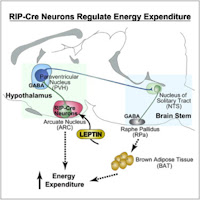
An important new paper on the methodology for doing gene co-expression network analysis was recently published in PLoS ONE by Kumari et al. (2012). The paper is entitled "Evaluation of Gene Association Methods for Coexpression Network Construction and Biological Knowledge Discovery".
An important new paper on the methodology for doing gene co-expression network analysis was recently published in PLoS ONE by Kumari et al. (2012). The paper is entitled "Evaluation of Gene Association Methods for Coexpression Network Construction and Biological Knowledge Discovery".The authors perform a comparative analysis of several different approaches for constructing co-expression networks.
____________________________________________________
Abstract:
Constructing coexpression networks and performing network analysis using large-scale gene expression data sets is an effective way to uncover new biological knowledge; however, the methods used for gene association in constructing these coexpression networks have not been thoroughly evaluated. Since different methods lead to structurally different coexpression networks and provide different information, selecting the optimal gene association method is critical.
Methods and Results: In this study, we compared eight gene association methods – Spearman rank correlation, Weighted Rank Correlation, Kendall, Hoeffding’s D measure, Theil-Sen, Rank Theil-Sen, Distance Covariance, and Pearson – and focused on their true knowledge discovery rates in associating pathway genes and construction coordination networks of regulatory genes. We also examined the behaviors of different methods to microarray data with different properties, and whether the biological processes affect the efficiency of different methods.
Conclusions: We found that the Spearman, Hoeffding and Kendall methods are effective in identifying coexpressed pathway genes, whereas the Theil-sen, Rank Theil-Sen, Spearman, and Weighted Rank methods perform well in identifying coordinated transcription factors that control the same biological processes and traits. Surprisingly, the widely used Pearson method is generally less efficient, and so is the Distance Covariance method that can find gene pairs of multiple relationships. Some analyses we did clearly show Pearson and Distance Covariance methods have distinct behaviors as compared to all other six methods. The efficiencies of different methods vary with the data properties to some degree and are largely contingent upon the biological processes, which necessitates the pre-analysis to identify the best performing method for gene association and coexpression network construction.